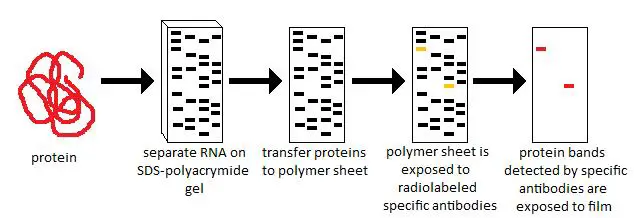
Its various formats of membranes were used to study the role of TRAF4 in modulating tight junctions and promoting cell migration, pheromonal ligands, protein synthesis initiation for MHC class I peptides, and the regulatory effect of PD-1 on IgA selection and the composition of gut microflora. The number 1 provider of nitrocellulose membranes is GE Healthcare, with the brand Amersham Hybond or Whatman Protran. Its immobilon PVDF membranes, for example, have been used to investigate choroid plexus organoids, Cox-2 and mPGES-1 expression in mouse bone marrow–derived dendritic cells, NLRP1B inflammasome, the involvement of L1 retrotransposon during cellular senescence and the importance of the regulation of matrimony levels to study the oocyte-to-embryo transition in Drosophila. MilliporeSigma is the primary provider of PVDF membranes. Table 3 lists the major suppliers for both types of membranes. It's a particular problem with blots exposed on film where there can be saturation of the film exposure, maybe less so with more recent fluorescence-based approaches.Two types of membranes are used for the protein transfer during the Western blotting: polyvinylidene difluoride (PVDF) and nitrocellulose membranes. That difference among biologic replicates is the variability against which you should statistically evaluate any differences you observe.įurthermore, quantifying Western blots can have serious pitfalls, depending on how it's done. You will almost always find that the variability among experiments on biologic replicates is much greater than the within-experiment errors from loading, blotting, exposing, and reading the samples. You really have to do the whole experiment several times, starting from fresh cells or tissues, to have a biologically reliable result. Please don't believe any results based on a single biologic replicate. And re-running the same protein extracts on another gel doesn't count as a biologic replicate that would simply account for the random vagaries of gel loading, antibody binding, film exposure conditions, etc. Re-reading the densitometry results would help account for some of the random vagaries of densitometry, but it would not get at the more fundamental and potentially much larger source of error: the differences among true biologic replicates of the same experiment. The main problem you have is what you fear: you can't do proper statistical analysis on a single observation. Please excuse me if the questions are too obvious or foolish. I'm a medical student and as is apparent, my stats are a little weak. Values for the control group are going to be 1. If I read them thrice, since these are normalized values, all three.Surely mention in my report if I did so :) ). So can I use them as three observations (I'll Does that matter? ANOVAs and any post hoc test would require a spread of values, right?ĭo i read the densitometry thrice so as to get a mean and an SD?Įven if I read thrice, isn't it that ANOVA assumes them to be in normal distribution? I guess for that we need more than 20 observations.Ĭan I say comment upon significance in differences when I have just one observation in each group?Įven if I read the same blot thrice, I expect to have slightlyĭifferent values. Since I've managed to run the blot just once, it is a single observation and I don't have a mean and SD. What kind of a statistical test should i put in? Do stats even work for single observations? I have normalized the densitometry values to the control group.This means that protein expression in the control is taken as 1 and that of the rest are shown relative to this control group. Technically, the blot's to be repeated 3 times but I've managed to run it just once. The null hypothesis being, that the protein expression is same in all the groups. I have a control group and 6 different test groups. I am doing a student project and have to analyse the western blot results.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
